CGT Ophthalmology

Cell and Gene Therapy (CGT) Ophthalmology Services
Pharmaron provides ocular disease models to help evaluate cell and gene therapy (CGT) products’ efficacy in disorders affecting the eye. Combining reliable preclinical disease models with multimodal imaging, histology and comprehensive biological assays specifically designed for CGT products can progress the pipeline forward. Our scientists are at the forefront of ophthalmic therapeutic development.
We have access to board-certified veterinary ophthalmologists and onsite support from attending veterinarians to ensure the study is conducted in compliance and in accordance with animal welfare regulations. Pharmaron has experience with many key ocular disease models to help evaluate your drug’s efficacy, which includes sophisticated CGT product delivery in ocular compartments, comprehensive evaluation and imaging for both the front and back of the eye.
Capabilities
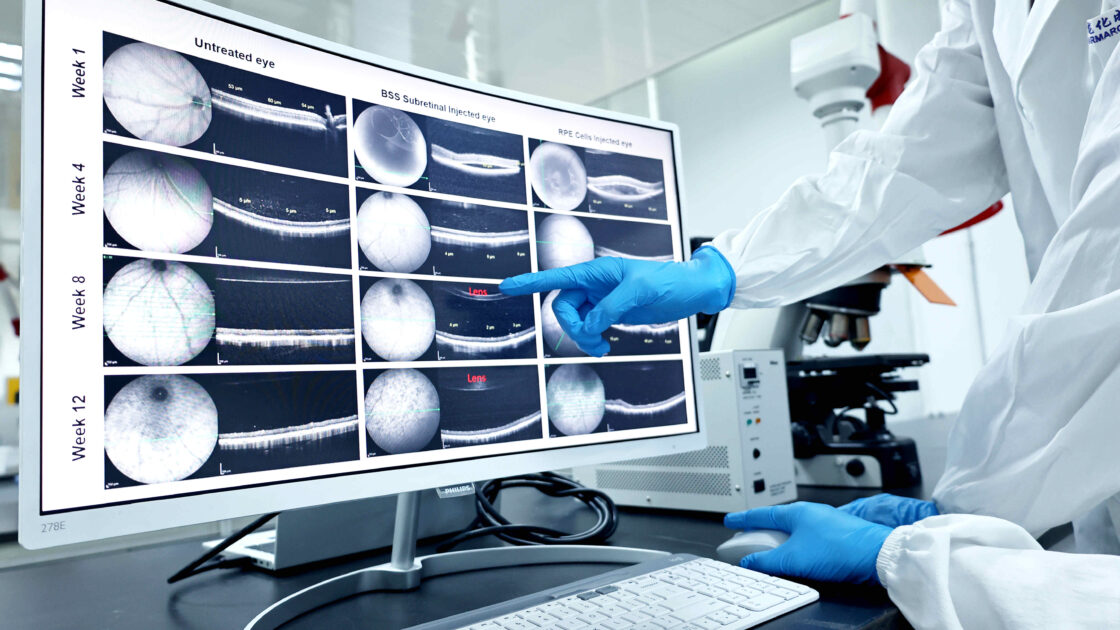
Ocular Disease Models
- Glaucoma
- Choroidal Neovascularization (CNV)
- Age-Related Macular Degeneration (AMD)
- Keratoconjunctivitis Sicca (KCS)
- Allergic conjunctivitis
- Anterior uveitis
- Infectious keratitis
- Neuroprotection
- Corneal wound healing
- Diabetic macular edema

Ocular Toxicology
Pharmaron’s ocular toxicology studies can be conducted under GLP (histopathology and clinical pathology) or non-GLP conditions. Our team has experience using small molecules, biologics, cell therapy, gene therapy, implants and transplants and works with rodents and non-rodents.